Myotonic Dystrophy (DM)
Myotonic Dystrophy (DM)
What is myotonic dystrophy (DM)?
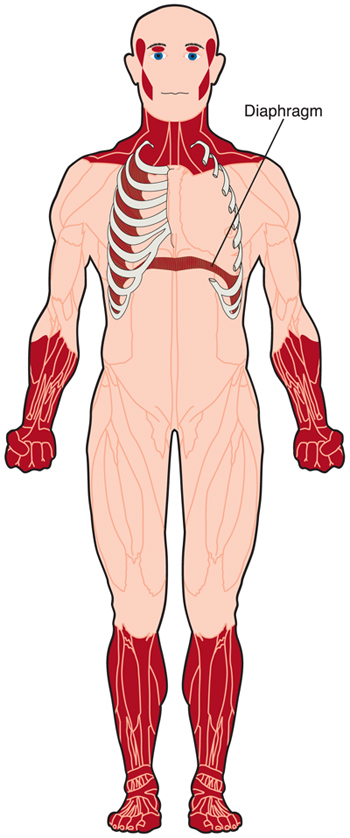 Myotonic dystrophy (DM) is a form of muscular dystrophy that affects muscles and many other organs in the body.
Myotonic dystrophy (DM) is a form of muscular dystrophy that affects muscles and many other organs in the body.
The word “myotonic” is the adjectival form of the word “myotonia,” defined as an inability to relax muscles at will.
The term “muscular dystrophy” means progressive muscle degeneration, with weakness and shrinkage of the muscle tissue.
Myotonic dystrophy often is abbreviated as “DM” in reference to its Greek name, dystrophia myotonica. Another name used occasionally for this disorder is Steinert disease, after the German doctor who originally described the disorder in 1909.
What causes DM?
DM is divided into two types.
Type 1 DM (DM1), long known as Steinert disease, occurs when a gene on chromosome 19 called DMPK contains an abnormally expanded section located close to the regulation region of another gene, SIX5.
Type 2 DM (DM2), recognized in 1994 as a milder version of DM1, is caused by an abnormally expanded section in a gene on chromosome 3 called ZNF9. DM2 was originally called PROMM, for proximal myotonic myopathy, a term that has remained in use but is somewhat less common than the term DM2.
The expanded sections of DNA in these two genes appear to have many complex effects on various cellular processes. In both DM1 and DM2, the repeat expansion is transcribed into RNA but remains untranslated in protein.
These conditions are some of the most common forms of adult-onset muscular dystrophy. DM is the most common muscular dystrophy among adults of European ancestry. The prevalence of DM is about 10 cases per 100,000 individuals.1,2,3,4 Among nonwhite populations, DM1 is uncommon or rare.5,6,7,8 Reports from Europe suggest the prevalence of DM2 is similar to that of DM1.
What are the symptoms of DM?
DM causes weakness of the voluntary muscles, although the degree of weakness and the muscles most affected vary greatly according to the type of DM and the age of the person with the disorder.
Myotonia, the inability to relax muscles at will, is another feature of DM. For example, it may be difficult for someone with DM to let go of someone's hand after shaking it.
As the disease progresses, the heart can develop an abnormal rhythm and the heart muscle can weaken. The muscles used for breathing can weaken, causing inadequate breathing, particularly during sleep.9
In addition, in type 1 DM, the involuntary muscles, such as those of the gastrointestinal tract, can be affected. Difficulty swallowing, constipation, and gallstones can occur.10,11 In females, the muscles of the uterus can behave abnormally, leading to complications in pregnancy and labor.12,13
The development of cataracts (opaque spots in the lenses of the eyes) relatively early in life is another characteristic of DM, in both type 1 and type 2.14
Overall intelligence is usually normal in people with DM but learning disabilities and an apathetic demeanor are common in the type 1 form.15 In congenital DM1, which affects children from the time of birth, there can be serious impairment of cognitive functioning. These children also may have problems with speech, hearing,16 and vision fatigue.
Generally, the earlier DM1 begins, the more profound the symptoms tend to be. For more, see Signs and Symptoms.
DM2 has a better overall prognosis than DM1. The symptoms are often relatively mild and progress slowly. DM2 rarely occurs during childhood, and there is no known congenital-onset form of DM2.
What is the progression of DM?
The progression of DM varies greatly among individuals, but in general, symptoms progress gradually. Life expectancy is clearly reduced for patients with congenital DM1 and is likely reduced for patients with childhood DM1 and classic (adult-onset) DM1.
The most common type of DM1 — the adult-onset form — begins in adolescence or young adulthood, often with weakness in the muscles of the face, neck, fingers, and ankles. The weakness is slowly progressive for these and eventually other muscles.
When DM1 begins earlier in life than adolescence — the congenital-onset and childhood-onset forms of the disease — it may be quite different in progression from the adult-onset type. Children with congenital-onset DM1, once they survive the crucial neonatal period of respiratory muscle weakness with the help of assisted ventilation, usually show improvements in motor and breathing functions. They may have cognitive impairment, delayed speech, difficulty eating and drinking, and various other developmental delays.15
The childhood-onset form of DM1, before the age of 10, is more often characterized by cognitive and behavioral abnormalities than by physical disabilities, such as intellectual impairment, attentional deficits, executive dysfunction, anxiety, and mood disorders.17, 18, 19 Eventually, muscle symptoms develop, to varying degrees.
DM2 is, in general, a milder disease than type 1. It does not appear to have a congenital-onset form and rarely begins in childhood.
In contrast to type 1 DM, the muscles affected first in DM2 are the proximal muscles — those close to the center of the body — particularly around the hips. However, some finger weakness may be seen early as well. The disorder progresses slowly, but mobility may be impaired early because of weakness of the large, weight-bearing muscles.
What is the status of research on DM?
Identification of the genetic mutations underlying DM1 and DM2, and understanding at least in part how the mutations cause disease, has opened up avenues for therapy development in DM.
Most of the strategies currently in development aim to block the harmful effects of the expanded DNA in the DMPK gene (type 1) or the ZNF9 gene (type 2). For more, see Research, In Focus: Myotonic Dystrophy, and particularly DM Research: Seeking to Free Proteins from a ‘Toxic Web.’
Download our Myotonic dystrophy (DM) Fact Sheet
References
- Understanding Neuromuscular Disease Care. IQVIA Institute. Parsippany, NJ. (2018).
- Magee, A. & Nevin, N. C. The Epidemiology of Myotonic Dystrophy in Northern Ireland. Public Health Genomics (2003). doi:10.1159/000016209
- Siciliano, G. et al. Epidemiology of myotonic dystrophy in Italy: Re-apprisal after genetic diagnosis. Clin. Genet. (2001). doi:10.1034/j.1399-0004.2001.590508.x
- Norwood, F. L. M. et al. Prevalence of genetic muscle disease in Northern England: In-depth analysis of a muscle clinic population. Brain (2009). doi:10.1093/brain/awp236
- Lotz, B. P. & van der Meyden, C. H. Myotonic dystrophy. Part I. A genealogical study in the northern Transvaal. S Afr Med J (1985).
- Ashizawa, T. & Epstein, H. F. Ethnic distribution of myotonic dystrophy gene. The Lancet (1991). doi:10.1016/0140-6736(91)90659-D
- Hsiao, K. M. et al. Epidemiological and genetic studies of myotonic dystrophy type 1 in Taiwan. Neuroepidemiology (2003). doi:10.1159/000071191
- Goldman, A., Ramsay, M. & Jenkins, T. Ethnicity and myotonic dystrophy: A possible explanation for its absence in sub-Saharan Africa. Ann. Hum. Genet. (1996). doi:10.1111/j.1469-1809.1996.tb01172.x
- Culebras, A. Sleep and neuromuscular disorders. Neurologic Clinics (2005). doi:10.1016/j.ncl.2005.08.004
- Hageman, A. T. M., Gabreëls, F. J. M., Liem, K. D., Renkawek, K. & Boon, J. M. Congenital myotonic dystrophy; a report on thirteen cases and a review of the literature. J. Neurol. Sci. (1993). doi:10.1016/0022-510X(93)90072-7
- Bellini, M. et al. Gastrointestinal manifestations in myotonic muscular dystrophy. World Journal of Gastroenterology (2006). doi:10.3748/wjg.v12.i12.1821
- R.N., S. & T.B., M. Pregnancy with myotoaic dystrophy. course, complications and management. Obstet.Gynec. (1971).
- Sarnat, H. B., O’connor, T. & Byrne, P. A. Clinical Effects of Myotonic Dystrophy on Pregnancy and the Neonate. Arch. Neurol. (1976). doi:10.1001/archneur.1976.00500070001001
- Arsenault, M. E. et al. Clinical characteristics of myotonic dystrophy type 1 patients with small CTG expansions. Neurology (2006). doi:10.1212/01.wnl.0000208513.48550.08
- Roig, M., Balliu, P. R., Navarro, C., Brugera, R. & Losada, M. Presentation, clinical course, and outcome of the congenital form of myotonic dystrophy. Pediatr. Neurol. (1994). doi:10.1016/0887-8994(94)90104-X
- Van Vliet, J. et al. Hearing impairment in patients with myotonic dystrophy type 2. Neurology (2018). doi:10.1212/WNL.0000000000004963
- Udd, B. & Krahe, R. The myotonic dystrophies: Molecular, clinical, and therapeutic challenges. The Lancet Neurology (2012). doi:10.1016/S1474-4422(12)70204-1
- Ekström, A. B., Hakenäs-Plate, louise, Tulinius, M. & Wentz, E. Cognition and adaptive skills in myotonic dystrophy type 1: A study of 55 individuals with congenital and childhood forms. Dev. Med. Child Neurol. (2009). doi:10.1111/j.1469-8749.2009.03300.x
- Douniol, M. et al. Psychiatric and cognitive phenotype of childhood myotonic dystrophy type 1. Dev. Med. Child Neurol. (2012). doi:10.1111/j.1469-8749.2012.04379.x
