
Caution, Preparation and Teamwork Lead to the Best Pregnancy Outcomes in Women with Neuromuscular Diseases
When Emma Ciafaloni was preparing to become a neuromuscular disease specialist in the 1990s, and even when she directed the MDA neuromuscular disease clinic at Duke University in the early 2000s, she was struck by the lack of information she could offer patients who wanted to become pregnant.
“I really have an interest in women and neuromuscular diseases, in what we can do better for them in dealing with their neuromuscular disease and also with pregnancy,” says Ciafaloni, now at the University of Rochester Medical Center, where she sees patients in the MDA clinic and has received MDA research support.
“I’m very interested in how to best care for patients. I’m not the one who’s going to discover the treatment for FSHD [facioscapulohumeral muscular dystrophy] or myotonic dystrophy, but I’m very interested in good standards of care.”Ciafaloni’s interest ultimately led her to collaborate with several colleagues in the departments of neurology and obstetrics and gynecology to conduct a study of pregnancy in women with FSHD. The results were published in 2006 in the journal Neurology.
 Ciafaloni’s interest ultimately led her to collaborate with several colleagues in the departments of neurology and obstetrics and gynecology to conduct a study of pregnancy in women with FSHD. The results were published in 2006 in the journal Neurology.
Ciafaloni’s interest ultimately led her to collaborate with several colleagues in the departments of neurology and obstetrics and gynecology to conduct a study of pregnancy in women with FSHD. The results were published in 2006 in the journal Neurology.
The researchers administered questionnaires to, and reviewed the medical records of, 38 women with FSHD. On the whole, pregnancy outcomes were good in this group, although the rates of operative deliveries (Caesareans and forceps deliveries) and babies with low birth weights were higher than the national average.
About one in four of the women surveyed reported worsening of FSHD symptoms that for the most part did not resolve after childbirth. The most common problems were, in order of frequency: worsening of generalized weakness, frequent falling, difficulty carrying the newborn due to worsening shoulder weakness, worsening or new-onset pain, and difficulty carrying the newborn due to worsening of leg weakness.
Despite some childbirth complications and possible permanent worsening of their FSHD, 90 percent of the women said they would choose pregnancy again.
Eva Pressman, director of maternal-fetal medicine at the University of Rochester, collaborated with Ciafaloni and others on the FSHD study. “The important thing about our study was that the outcomes were really good,” she says. “It’s important for patients to know that just because they have an underlying disorder, that doesn’t mean that pregnancy is contraindicated. With appropriate monitoring and going into the pregnancy with your eyes open as to what the risks might be, I think most patients can be quite successful.”
But not all neuromuscular disorders, let alone all women or all babies, are the same. And even with conditions like FSHD, where the odds of having a successful outcome appear to be good, there are steps women can take to improve them.
First, some red flags:
Cardiac involvement
Cardiac involvement can occur in many neuromuscular diseases and, if it’s severe, can be a major problem for women considering pregnancy.
Pregnancy leads to a significant increase in cardiac output, as well as a 50-percent increase in blood volume, so the heart does “much, much more work,” says Pressman. “And if you go into the pregnancy with a heart that’s functioning less than optimally, it will clearly deteriorate over the course of the pregnancy. That can lead to heart failure in the mother and endanger both the mother and the fetus, depending on when in the pregnancy those issues occur. The most common time for heart problems is the end of the second trimester or the beginning of the third trimester.”
Pressman strongly recommends that patients who are at risk for cardiac dysfunction have echocardiograms before pregnancy to evaluate their heart function and “really consider not becoming pregnant if they have significant cardiac dysfunction.”
And if they’re already pregnant and want to continue the pregnancy despite their heart problem? “Then you do the best you can,” Pressman says. “You can manage some of it with medications, you can keep their fluid status at an optimal level, and we can usually get them through to the point where the baby is viable. But often we deliver these patients early, to minimize the stress on the heart.”
Weak respiratory muscles
Weakness of the respiratory muscles can also be a problem for pregnant women with muscle disease. As the pregnancy progresses, the work of breathing becomes harder, so any existing impairment can become more problematic.
“We see women who have reasonable respiratory function prior to pregnancy that deteriorate as the pregnancy progresses,” Pressman says. They generally recover to their pre-pregnancy level after delivery, she notes, but they may require extra support (such as noninvasive assisted ventilation) during the pregnancy.
Says Pressman, “We often end up delivering a little bit early, because the respiratory impairment only gets worse towards the end of pregnancy, and sometimes the safest thing is to not be pregnant anymore.”
Unstable autoimmune disease
It’s critical that women with autoimmune disease are medically stable and on a stable regimen of medications for at least six to 12 months before they try to get pregnant, says Hannah Briemberg, a neurologist and assistant professor in the neuromuscular diseases unit at the University of British Columbia (Canada) in Vancouver.
The neuromuscular autoimmune diseases in MDA’s program — polymyositis, dermatomyositis, myasthenia gravis and Lambert-Eaton myasthenic syndrome — are generally treated with immunosuppressive drugs and sometimes other medications.
In a paper she published in 2007 in the journal Seminars in Neurology, Briemberg found that pregnancy does not appear to alter the long-term outcome of myasthenia gravis (MG) but that, if the disease is not yet stable before conception, there is more risk that it will worsen during pregnancy.
A phenomenon that can occur in MG but does not seem to occur in other autoimmune diseases is the temporary transmission of the disease to the baby, causing him or her to be born floppy and possibly with swallowing or breathing difficulties. (The phenomenon occurs because the antibodies the mother’s immune system produces that weaken her own muscles can cross the placenta and weaken the baby’s muscles, at least temporarily.)
That doesn’t seem to occur as often as it used to, Briemberg says, something she attributes to better MG care and mothers whose disease is better controlled during pregnancy.
Women with polymyositis or dermatomyositis also need to have their disease under control, preferably before getting pregnant. Briemberg found that women with myositis who were in remission at the time of their pregnancy and delivery did not appear to be at risk for obstetric complications. However, she found, women with active disease at the time of their pregnancies had an increased incidence of spontaneous abortion, premature delivery and low-birth-weight infants. The highest risk was associated with new-onset disease during the first trimester of pregnancy. In these women, the rate of fetal deaths was very high (although the number of women studied was small).
Although there are risks associated with the use of disease-controlling medications during pregnancy, Briemberg advises that these risks are, in general, preferable to the risk of being pregnant and having an uncontrolled autoimmune disease.
Corticosteroid drugs, such as prednisone, are often prescribed for autoimmune diseases, and generally appear to be fairly safe during pregnancy.
“Prednisone doesn’t cross the placenta very well,” says Eva Pressman, “so it’s one of the safer medications to use during pregnancy, because most of it is going to the mother, with very little of it getting to the baby.” It’s not entirely without risk, however. Prednisone and related medications can interfere with the growth of the fetus and have been associated with premature rupture of the amniotic sac, possibly by interfering with collagen formation, she notes.
Pressman adds that it can be impossible to determine whether problems are from the effects of prednisone or the effects of the mother’s underlying disease.
Another issue with corticosteroids is that, if they’re given for a long period of time, they can suppress the function of the adrenal glands, which normally pump out high levels of the stress-coping hormone cortisol during labor and delivery. Therefore, in general, doctors recommend that women who have been on prednisone or other corticosteroids be given intravenous corticosteroids (hydrocortisone) during labor and delivery.
Immunosuppressant medications that aren’t corticosteroids pose different types of concerns.
None of these medications have been systematically studied in pregnant women, Briemberg notes, so most of the data comes from animal experiments or data collected from women who happened to get pregnant while taking one of them.
“At this point, the clinical data and experience suggest that prednisone, azathioprine and IVIG [intravenous immunoglobulins] are unlikely to pose any significantly increased risk of fetal malformation,” Briemberg says. “There is not enough data on other immunosuppressive medications to know if they are safe in pregnancy, so most clinicians will recommend coming off these other medications prior to trying to conceive.”
Myotonic dystrophy — a special case
Most neuromuscular diseases affect mainly the voluntary muscles (in the limbs, trunk, head, face, and swallowing and breathing structures). The heart, though not a voluntary muscle, also is affected in many neuromuscular diseases.
But myotonic dystrophy (MMD) affects not only the voluntary muscles and the heart, but the involuntary, or “smooth,” muscles that line the hollow organs, such as the gastrointestinal tract, urinary tract, uterus and vagina.
Abnormalities of uterine and vaginal muscle function (either weakness or myotonia, the inability to relax muscles) can have severe adverse effects on labor and delivery.
“If the uterus is actually affected, then labor may not progress well,” Pressman says, “and you may not be able to have a vaginal delivery. We can try to alter that with oxytocin [a labor-stimulating hormone] or other medications, but if you can’t make the uterus contract, then you would need a C-section [Caesarean] to deliver.”
In addition, people with MMD are especially sensitive to pain-relieving medications and can have abnormal reactions to anesthesia, so there are additional worries on this account.
A very severe form of MMD called congenital MMD can occur in offspring of women with type 1 MMD who may themselves be minimally affected. So far, this phenomenon has not been seen in type 2 MMD. (Type 1 MMD is caused by an expansion of DNA on chromosome 19, while type 2 MMD, a similar disease, is caused by an expansion of DNA on chromosome 3.)
Babies with congenital MMD can be born very floppy, with respiratory impairment and sucking and swallowing difficulties, for which the obstetric and pediatric teams must be prepared.
Normally, babies swallow some of the mother’s amniotic fluid, the watery substance that surrounds the baby in the uterus. But a baby with congenital MMD can have so much swallowing impairment that excess amniotic fluid accumulates, further endangering mother and baby.
Excess amniotic fluid, called “polyhydramnios,” can cause premature rupture of the membranes and premature onset of labor, sometimes before the baby is ready to be born. If the membranes don’t rupture prematurely, the uterus can become so distended that the mother’s breathing is impaired and blood vessels can be compressed. Excess bleeding after delivery (postpartum) is also associated with an over-distended uterus during the pregnancy.
“If you have a baby with limited swallowing ability and you have polyhydramnios, and you’ve overstretched the uterus because of that, then even a normal uterus doesn’t contract well,” Pressman says. With a uterus affected by MMD, she notes, “your risk of postpartum hemorrhage is much higher.”
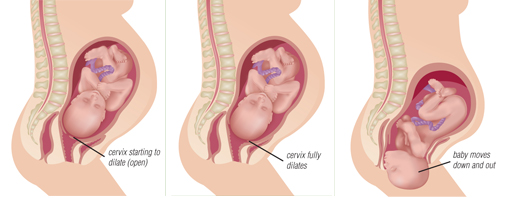
Planning Ahead: Five P’s
Traditionally, obstetricians and midwives have thought about pregnancy, labor and delivery in terms of three p’s: the passenger (baby), the passageway (the mother’s bone structure and soft tissues of the birth canal), and the powers (the involuntary contractions of the uterus and the voluntary pushing efforts of the mother’s abdominal muscles).
To this classical way of looking at things, two more p’s can be added: pain management and pregnancy-disease interaction.
Almost any pregnant woman with a neuromuscular disease can have difficulty with at least two of the three p’s, the passageway and the powers. And, if those difficulties are serious, they can affect the passenger as well.
In addition, pain management can pose some special challenges for some women with neuromuscular disease, and sometimes pregnancy can have some long-term adverse effects on disease progression.
Thinking of pregnancy and delivery in terms of the five P’s can help women with neuromuscular disease plan ahead.
The passageway for the passenger
Several factors can affect the passageway.
“The size of the mother’s pelvis is clearly important and can affect a woman’s ability to have a safe vaginal delivery,” says Eva Pressman.
David Colombo, a maternal-fetal medicine specialist at Ohio State University Medical Center in Columbus, has seen small “juvenile-type” pelvises in several of his patients with spinal muscular atrophy (SMA). He prefers to do a C-section in these cases.
Says Pressman, “It’s not required to have a C-section just because of a small pelvic diameter, but it would be the kind of thing where you would have the discussion ahead of time, watch the progress of labor, and perhaps not wait through three or four days of labor before deciding enough is enough.”
Sometimes, contractures (joints frozen in one position) in the hips, knees or spine can pose an impediment to delivery and may lead a physician to recommend a C-section.
Spinal curvatures — and sometimes the surgical procedures performed to correct them (spinal fusions, often involving insertion of instruments, such as rods and screws) — can be a problem.
The growing uterus can press on an abnormally shaped or fused spine, possibly causing loss of sensation, increased weakness or even paralysis. 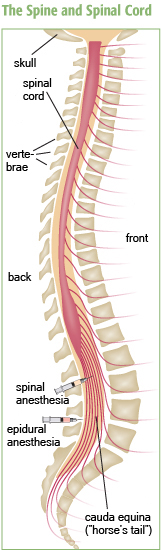
Because of increased pressure and strain on the lower back during pregnancy, compression of the cauda equina (the “horse’s tail” formation of spinal nerves in the lumbar and sacral areas) does occasionally occur, even in healthy women, says Hannah Briemberg. The risk may be greater in neuromuscular diseases, such as spinal muscular atrophy. However, notes Briemberg, “It’s still an extremely rare complication.” She cautions that low back pain affects almost everyone during pregnancy, and the vast majority of the time, it does not mean anything is seriously wrong.
The powers
The first stage of labor is one in which the cervix (the neck of the uterus) dilates so that the baby has room to leave the uterus during the second stage, when the baby is pushed out.
The first stage is entirely the result of smooth muscle uterine contractions, which are involuntary. Women with neuromuscular diseases (with the exception of myotonic dystrophy) generally don’t have any special problems with this stage of labor.
However, almost all women with neuromuscular diseases, (including those with myotonic dystrophy) have voluntary muscle weakness and have some difficulties with the second stage of labor.
The uterus continues to contract during the second stage, helping to expel the baby. However, the mother’s voluntary efforts, by contracting her abdominal muscles, add force in pushing the baby out in a timely manner.
“It’s more difficult to push when the pushing stage comes, if you don’t have much strength to begin with,” says Briemberg. “In women who have prolonged labors, having some voluntary muscle strength to push could help.”
That doesn’t necessarily mean women with neuromuscular disease have prolonged or difficult labors or that operative interventions will be needed, but it does put women with these disorders at increased risk for needing them.
“As far as the actual labor and delivery are concerned, most of the force is generated by the uterus, which is smooth muscle,” Pressman says. “The uterus is quite capable of expelling a baby with no effort at all on the mother’s part. So, even though we encourage women to push, they don’t always need to push. Sometimes that additional effort is helpful, but much of the time, it’s not needed.” However, she notes, if “it’s a very snug fit,” or if the uterus itself is not functioning properly, physicians may intervene.
Pain management
Spinal abnormalities or the effects of spinal surgery can make regional anesthesia, such as spinal or epidural blocks, difficult. These types of anesthesia may not work as well in patients with significant spinal abnormalities or who have had instruments such as metal rods inserted.
 “It’s not necessarily contraindicated to get these types of anesthesia, but they may not work as well,” says Pressman. “Any time you’ve operated on the spine, there can be scarring, and so getting the medication to spread properly is difficult. If the space between the spinal elements is narrowed because of a change in diameter of the spine, then it can be hard to get the anesthetic in as well.”
“It’s not necessarily contraindicated to get these types of anesthesia, but they may not work as well,” says Pressman. “Any time you’ve operated on the spine, there can be scarring, and so getting the medication to spread properly is difficult. If the space between the spinal elements is narrowed because of a change in diameter of the spine, then it can be hard to get the anesthetic in as well.”
The same barriers apply to spinal or epidural anesthesia. The difference in these is in the exact placement of the anesthesia-delivering catheter (see illustration "The Spine and Spinal Cord").
On the other hand, general anesthesia (inhaled anesthesia), which is an alternative to epidural or spinal anesthesia for C-section deliveries, poses other hazards for some women with neuromuscular disease.
Women with central core disease, for example, are at very high risk for a severe adverse reaction called “malignant hyperthermia,” when given certain inhaled anesthetics. It’s very important that the obstetric team know about this risk beforehand, so they can use alternative anesthetic agents.
And, once again, the woman with myotonic dystrophy is at high risk of an adverse reaction (though not necessarily malignant hyperthermia) to general anesthesia and, in her case, also to the pain-killing medications that are sometimes given intravenously during labor.
“The big concern is for the anesthesiologist and obstetrician to be aware that they might be a little bit more sensitive to these intravenous medications that are given during labor and maybe to not dose quite as heavily as they would in a woman who didn’t have myotonic dystrophy,” Briemberg says.
If a woman needs general anesthesia during a surgical delivery, she’s generally “intubated,” meaning the surgical team puts a tube down her trachea, and a ventilator breathes for her during the surgery. In the average person, the tube can be removed almost immediately after the delivery, and she can breathe on her own without difficulty.
However, women with very weak respiratory muscles, such as someone with spinal muscular atrophy or uncontrolled myasthenia gravis, may have great difficulty resuming normal respirations after having been intubated, even for a short time.
Fearing this complication, some women have opted for an unusual way to manage pain relief during a C-section — local anesthesia.
David Colombo, who delivered Stacy Wiparina’s babies by C-section, used local anesthesia both times. “We have a whole protocol that involves 100 cc of lidocaine,” Colombo says. “I numb the skin, and then we make our incision. Then you go into the fascia [layer of tissue just under the skin], and you incise that, and then you numb the peritoneum [deeper layer of tissue] over the uterus. They don’t feel it when you cut into the uterus; there’s no sensation there. Actually, people tolerate it very well. If you take your time and do it right, it’s just the same as a spinal. Nobody really complains. And you don’t have to worry about intubation.”
Eva Pressman, however, isn’t as sure about using local anesthesia for a C-section. “I’ve seen it done a couple of times,” she says. “You can numb the skin and the fascial layers very effectively, but you can’t numb the peritoneum very well. Once you get into the abdominal cavity, you get pretty uncomfortable. That being said, I worked with an older obstetrician 20 years ago who had three C-sections herself, all under local anesthesia, because that’s what she wanted. It would not be my recommendation, but if you get the right sort of person, you can do some things that other people might not be able to tolerate.”
Pregnancy and disease progression
“I think the big thing is that, almost as a rule, you have to expect that you’re going to get a little bit weaker during the pregnancy and, unfortunately, that you’re probably not going to bounce back after the delivery,” Briemberg says. “Why they get weaker, I don’t think we know. It doesn’t happen to everyone, but it certainly does happen to a significant number, so I think they have to be prepared for that.”
Even if weakness doesn’t increase permanently, the pregnant woman with a neuromuscular disease is almost certain to experience a temporary ratcheting up of her disability level, says Pressman. “Pregnancy is a time in your life when you’re gaining more weight than you would normally gain in a short period of time,” she notes. “So, if you have enough muscle strength to ambulate and carry on your routine activities, but you gain 20 or 30 percent more weight over the course of the pregnancy, you might not be able to continue those sorts of things. You also change your center of gravity, so you use different muscles to support and balance yourself, and your weakness might become more evident.”
Teamwork makes the difference
Coordination and communication among the pregnant woman, her neurologist and a team experienced in high-risk obstetrics (such as can be found at most major medical centers) are essential to a good outcome, experts agree. In addition, meeting with a genetic counselor can help couples assess the risk of passing a genetic disorder to a child. Carrier testing and prenatal diagnosis are available for many diseases. (See “The Pain and Promise of Prenatal & Newborn Genetic Diagnosis”.)
"Care that is well coordinated among services is safe care, and care that is done in isolation can be less safe,” Pressman says. “It’s really important that the different care providers communicate with one another and that the patient tells the same things to different providers. I’ve had patients who have come to me and who’ve sort of downplayed their illness. Then I get records from their neurologist, and it turns out they’ve had many more complications than they’ve been willing to share with me.”
Pressman recommends that, ideally, women with a neuromuscular disorder have a consultation with a physician with expertise in complicated pregnancies before becoming pregnant. “Once you’re pregnant,” she notes, “there are fewer options.”
 Briemberg adds, “It’s important to have a pre-delivery anesthetic consult, in case you need general anesthesia.” If anything, she says, women with neuromuscular diseases “end up being safer than women that don’t have a neuromuscular disease,” if they have a preoperative anesthetic consult, because “everybody’s very in tune to all the issues and knows what agents can and can’t be used.”
Briemberg adds, “It’s important to have a pre-delivery anesthetic consult, in case you need general anesthesia.” If anything, she says, women with neuromuscular diseases “end up being safer than women that don’t have a neuromuscular disease,” if they have a preoperative anesthetic consult, because “everybody’s very in tune to all the issues and knows what agents can and can’t be used.”
Worth it
“My experience is that most people feel it was still worthwhile having the pregnancy,” says Briemberg. In fact, she says, it’s important for doctors to “be realistic,” while at the same time not causing women to worry too much.
“We’re all so anxious when we’re pregnant, about everything,” she says, “and I really try more than anything to be reassuring. For most of these women, it’s not going to have a huge impact on their disease. They’re going to have all the trials and tribulations of being a parent, but for the most part, the disease isn’t a huge additional factor.”
MDA Resource Center: We’re Here For You
Our trained specialists are here to provide one-on-one support for every part of your journey. Send a message below or call us at 1-833-ASK-MDA1 (1-833-275-6321). If you live outside the U.S., we may be able to connect you to muscular dystrophy groups in your area, but MDA programs are only available in the U.S.
Request Information