
The Pain and Promise of Prenatal and Newborn Genetic Diagnosis
Our son is wonderful,” says K.M., a 42-year-old San Francisco area events manager and the mother of a 7-year-old boy with Duchenne muscular dystrophy. “I couldn’t ask for anything more. He’s everything I ever wanted. Of course I wouldn’t want this disease, but he’s just such a great, sweet kid, and it’s been our hope and dream to give him brothers or sisters.
“When we found out our son had DMD — you just go through stages. His whole life passes through your head, and then you think, oh my God, there’s no way we could ever have more children; and then you accept that this is your reality.
“It was probably six to eight months later when we said we want to have more children, and we’ll use all the medical resources available to do it.”
In September, K.M., who asked not to be identified, gave birth to healthy twins, a girl and a boy, conceived using in vitro fertilization and preimplantation genetic diagnosis. After trying and failing to obtain healthy fertilized embryos from her own eggs, she and her husband ended up using an egg donor.
She estimates they spent $20,000 on in vitro fertilization (IVF), in which an egg and sperm are brought together in a laboratory dish and then the resulting embryos are implanted into the woman’s uterus. They spent another $5,000 for preimplantation genetic testing, and $30,000 for the donated eggs. None of that was covered by insurance.
K.M. was overjoyed when she found out she was pregnant, but the situation wasn’t without its intimidating aspects.
“I thought, I’ve got a son with MD; I just spent $50,000; and I’m carrying twins from an egg donor,” she recalls. “If I heard this story from someone else, I would think, ‘wow.’ But it’s just what you’re given.”
Test tube babies
The birth of Louise Brown in the United Kingdom in 1978, the first “test tube baby,” ushered in the era of IVF, allowing infertile couples to have biological children, with conception occurring outside the womb. Or they could use egg or sperm donors for certain types of infertility or to avoid a familial condition.
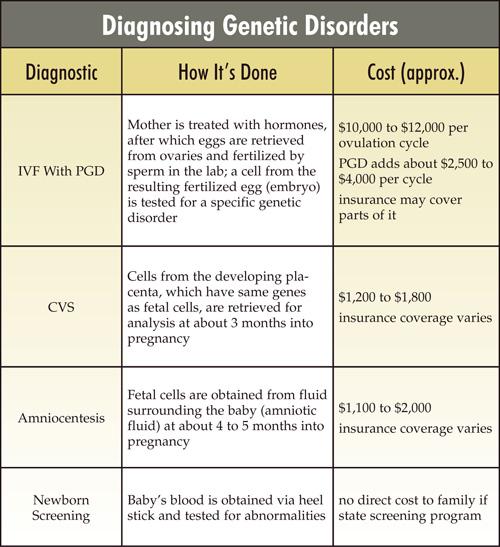
Preimplantation genetic diagnosis (PGD), available since the mid-1990s, requires IVF, followed by removal for analysis of one of the embryo’s cells two or three days after fertilization. At this stage, the embryo contains five to eight identical cells and theoretically sustains no damage as a result of having one, which the dividing cells will quickly replace, removed.
PGD is almost always done because a couple has a history of a genetic disease. An embryo carrying a disease-causing genetic mutation, identified when the sample cell is removed for analysis, won’t be implanted. Embryos diagnosed as free of the mutation will be implanted in the prospective mother’s uterus.
Fears of a slippery slope
As one might expect, not everyone is enthusiastic about the technology. Some argue that no fertilized egg should ever be discarded, no matter what its genetic makeup or stage of development.
And many disability activists view PGD as a form of genocide that will ultimately thin their ranks, undermine their goals and make life even harder for those born with disabling conditions.
If preimplantation genetic diagnosis has its desired effect — reducing the number of children born with disabling conditions — “it could change the way society views the disabled,” says a 2004 report by the Genetics and Public Policy Center at Johns Hopkins University in Baltimore.
At the same time, bioethicists have voiced concerns about a slippery slope between selecting embryos free of genes that cause serious disease and selecting those that meet parents’ desires for children whose DNA predicts high intelligence, mental stability or attractiveness.
In its 2004 report, the Hopkins center notes that societal norms could evolve to the point where parents might be pressured to use PGD, and to a society where aesthetic concerns, convenience or prejudice “supplant the inherent dignity due to every human being.”
There are also some practical concerns.
Is it safe?
Some physicians and scientists argue that IVF, which involves giving a woman high doses of hormones to produce as many eggs as possible (superovulation), followed by an invasive procedure to retrieve them from the ovaries, isn’t necessarily safe for the mother or the future baby. They also say that PGD may further jeopardize the offspring’s well-being.
The way IVF is practiced today involves putting several embryos into the uterus, with the hope that at least one will “take.” Quite often, more than one does, leading to twins, triplets or even higher numbers of multiples, with a corresponding increase in the complication rate for mothers and babies.
An Australian study of infants conceived through IVF and born between 1993 and 1997 found twice the rate of major birth defects compared to naturally conceived babies. The interpretation of these results is difficult, because the infertility or advanced maternal age that drives many couples to use IVF are risk factors in themselves. However, no one really knows whether there are other factors leading to birth defects, such as the medications used to induce superovulation, the freezing or thawing of embryos, or differences in the timing of fertilization compared to the natural process.
Is it reliable?
“Look, if you ever find a physician or scientist that tells you something in biology is 100 percent, I would fire them quick,” Mark Hughes, director of Genesis Genetics Institute in Detroit, tells couples. “We try to explain to the couple that what this test is doing is testing the smallest unit of life, one cell, and the smallest unit of inheritance, one gene, and the smallest part of a gene, oftentimes a single nucleotide [DNA element].
“The goal here is to decrease their odds, their risk, from 25 percent for most of these disorders all the way down to 3 percent or 4 percent or 2 percent, but not to zero.”
Hughes explains, “This is not math. In math, you have a precise science, and you can get precise answers. Just about the time we think we’ve got Mother Nature by the tail, she comes around and kicks us in our own, and reminds us that we’re not as smart as we think we are.”
Testing during pregnancy
Of course, all these concerns (with the exception of specific safety issues) pertain to any form of prenatal genetic testing, including chorionic villus sampling (CVS) and amniocentesis, both of which predate PGD.
CVS, available since the 1980s, in-volves taking a sample from the chorion, a membrane surrounding the fetus and containing fetal cells, about 10 to 12 weeks into the pregnancy. Any genetic test that can be performed on an embryo also can be performed on these cells, with the option to terminate the pregnancy if the tests predict a serious disease.
Amniocentesis involves withdrawing a small amount of fluid from the sac surrounding the fetus, from which fetal cells can be extracted and genetically analyzed. Amniocentesis is typically performed after 15 weeks of pregnancy, and there may be considerable turnaround time for the results of genetic testing for a disease. If results show an affected fetus, parents can consider a second-trimester termination, a highly distressing procedure for almost anyone.
Both of these techniques are more reliable at predicting a diagnosis than is PGD because there are far more cells to work with. With PGD, technicians examine just one cell per embryo. With CVS or amniocentesis, many cells from the same fetus can be tested.
They’re also simpler to perform, carry fewer risks for the mother and baby, and are far less expensive than PGD. But the consequences of getting an undesired result during pregnancy can be agonizing.
“Most couples,” says Mark Hughes, “look at this and say, ‘This [PGD] is better than our other alternatives. We’re just not interested in taking a chance. Until we have cures, this is a great thing.’”
Judy Yamada, an Arizona woman whose brother died of Duchenne MD in 1990, agrees. She went through IVF and PGD four times before achieving a pregnancy on the fifth, in January.
“It always seemed like he lived a normal life,” she says of her brother, Gary Luke. “He was in good spirits and he was smart and did well in school and had friends in school, but I think the frustration, as he got older, was harder and harder. He lived to 21. [Having a child with DMD] is not something I can consider.”
On the other hand, she says, “If I got pregnant and had [an affected fetus], that’s a hard choice. That’s why I didn’t like CVS. Once you’re pregnant for three months, it’s hard. I don’t want to make that choice.”
Testing newborns
While the goals of prenatal testing are prevention and preparation, the goal of postnatal testing is treatment. The case for widespread newborn screening is especially compelling if starting a treatment early is crucial to health or survival.
(At present, genetic tests for neuromuscular diseases are used primarily by people who already have a family history of a disease.)
States have been screening newborns for certain conditions, such as phenylketonuria (PKU), a metabolic abnormality that can be treated immediately with dietary measures, since the 1960s. But the burgeoning science of molecular genetics has increased exponentially the number of available tests since that time.
A few years ago, the U.S. Health Resources and Services Administration’s Maternal and Child Health Bureau commissioned the American College of Medical Genetics (ACMG) to study newborn screening and make recommendations about programs for it, which are under the control of each individual state.
The ACMG convened a Newborn Screening Expert Group, which included R. Rodney Howell, a professor of pediatrics at the University of Miami and chairman of MDA’s Scientific Advisory Committee, and published its findings in May 2006.
The panel concluded that, to be part of a newborn screening program, a disorder should be detectable by a reliable test 24 to 48 hours after birth; not ordinarily be detectable without the test; and benefit from early diagnosis and treatment. Twenty-nine conditions met these criteria. (Almost all are metabolic disorders, and none except carnitine uptake defect are in MDA’s program.)
Acid maltase deficiency (Pompe disease), an MDA-covered disorder, didn’t make the list, because neither an effective treatment nor a reliable screening test for it was available until after the ACMG report had gone to press. Last year, the Genzyme product Myozyme was approved as a treatment for Pompe’s.
“When this report came out, we didn’t have Myozyme, and we didn’t have appropriate testing,” Howell, a member of MDA’s Board of Directors, says. “Now we have an approved therapy. We just have to demonstrate that the test is ready for public health use.”
Some experts advocate screening newborns for diseases that don’t quite meet the ACMG criteria.
The case for SMA screening
“Newborn screening for spinal muscular atrophy (SMA) would be useful,” Howell says. In type 1 SMA, weakness of breathing and swallowing muscles begins in early infancy and usually leads to death within about two years. The less severe forms, types 2 and 3, begin later and allow a much longer life span.
In type 1 SMA, Howell notes, “the drop in motor neurons is precipitous after birth.” Whatever therapy is to be given has to be administered before this happens, he says.
Tom Prior, a professor and director of the Division of Molecular Pathology at Ohio State University, developed genetic testing for SMA several years ago, with MDA support, and is also a strong proponent of newborn screening for this disease.
“By the time patients are symptomatic, you may not have many motor neurons left,” Prior says. So far, there are no treatments for SMA that are clearly beneficial, but even experimental treatments, he says, may be futile unless affected babies can be identified early enough to benefit from them.
“If you want to evaluate the efficacy of phenylbutyrate or valproic acid,” he notes, naming two compounds now being tested in the disease, “you may not be able to assess that when patients are already sick.”
Prior says SMA newborn testing “is not ready for prime time yet” and requires a pilot program. But, he says, the “door is being opened.”
The case for DMD screening
“We’re staring down a barrel at antisense oligonucleotide and stop codon read-through therapies, as well as gene therapy,” Howell says, referring to several experimental treatments for DMD. “And it would be a tragedy not to be prepared.”
He expects some disease-modifying agents will be available for DMD soon, but that, as in SMA, they’ll have to be given early to have maximum benefit.
“You have to be ready with anything you have in the hopper, so you can prevent damage. You want to identify babies who appear normal but can benefit from treatment.”
Elevated creatine kinase, an enzyme that leaks out of damaged muscle cells, needs to be looked at as a screening tool, Howell says. But elevations of CK are common in the newborn period, perhaps because of injury during the birthing process.
“A two-tier test might well be needed for DMD,” Howell says. “If you screen early in infancy, you’ll get lots of falsely elevated CKs. So you have to take all those outside the normal range and do a second test, looking at DNA.”
Howell says parents can use information about DMD from a newborn screening test in making family planning decisions, but he doesn’t think that’s its main purpose.
“I would not be excited unless there were effective treatments coming,” he says. “For these new treatments, you want to identify patients very early.”
Caveats and concerns
It’s hard to argue with providing a timely, effective treatment for a newborn baby, but until those treatments are readily available, some experts warn, it’s also important to think about the potential downside of diagnosing a child before symptoms occur.
Nicole Johnson, a genetic counselor associated with the MDA clinic at Johns Hopkins University in Baltimore, studied the effects on parents of diagnosing DMD during the newborn period for her master’s degree thesis.
In 2001, she interviewed 12 out of the 58 families who’d been through this experience during a 12-year pilot MD newborn screening program that ended in 1997. She found mixed reactions. Although all of the parents felt that the newborn period wasn’t the appropriate time for an early diagnosis, no parent said that in retrospect they would have preferred a later, clinical diagnosis.
Some parents noted that having the diagnosis so early allowed them to adjust to it, so that by the time the child began asking his own questions, they felt prepared. Also mentioned were an appreciation of more time to make better decisions about career plans, child care options, and home renovations or purchases geared to the needs of a wheelchair user.
Some parents started their very young children on stretching exercises, swimming or chiropractic visits, with the hope of forestalling the effects of DMD. Seven of the 12 families surveyed said they’d changed their original reproductive plans based on knowing their son’s diagnosis.
But there were several concerns expressed by parents as well. Four parents described at least an initial period of feeling detached from their new baby once they got the diagnosis.
Others reported a feeling of chronic, bittersweet sorrow as they watched their child grow and develop. Each developmental milestone reached was greeted with excitement and at the same time with the knowledge that it probably wouldn’t be maintained for long.
“I grabbed the video camera when my child ran across the yard,” one parent told Johnson. “We try to cherish more moments than we would if we knew that he was healthy, which is a shame. But I think in the back of our minds, we know there are things we need to remember.”
Even foreknowledge of the risks to future children could be a double-edged sword, with some parents reporting that they felt alienated from the medical community if they didn’t subsequently use this knowledge to avoid having another affected child.
Information and support
Parents in Johnson’s study felt that two key elements were missing from their experience with early screening: a better informed consent process for the MD-specific portion of newborn screening, and an improved, more supportive practice for the disclosure of results.
Parents in this study felt that the most negative aspect of their experience with newborn screening for MD was the process through which they first heard about their son’s possible diagnosis. They often didn’t recall giving their consent for MD screening. Many parents had assumed that everything their baby was being tested for would be treatable and were shocked to find out this wasn’t the case.
When they were later told that a screening test had revealed that their sons might have muscular dystrophy, an untreatable condition, they experienced this as completely “out of the blue.”
In two families, she says, parents who didn’t even realize that their baby had been tested for MD were notified over the phone right before the July Fourth weekend that a screening test for MD was positive. They had to wait several days before further information was available.
“Any future newborn screening program for MD has to take into account a thoughtful protocol for disclosing results that provides the best support to the parents,” Johnson says. “Professionals have to understand that this is unlike any other newborn screening, where treatment begins soon after diagnosis. There were unique emotional needs of the parents who were learning of their son’s MD test results.”
Are we there yet?
Howell agrees that a lot more support is needed for parents of newborns whose screening tests cause concern.
“If a clerk calls and says, ‘Your baby has a positive test,’ the parent goes right to the Web and sees frightening information,” he says. “If a professional person calls, and says the screen is out of the usual range and the test needs to be repeated, you get a different message.”
He nevertheless remains enthusiastic about newborn testing. “Most people would like to know about a serious problem with their baby as soon as possible.”
Howell says parents of children with fragile X syndrome, the most common genetic form of mental retardation and one that so far has no treatment, are “organized and powerful” advocates for newborn screening.
“These parents say they spent two years trying to figure out what was wrong with their child, and in the meantime they had two more children with fragile X,” Howell says.
While he doesn’t think newborn screening for SMA and DMD should “start Monday morning,” it shouldn’t wait too long either. “Pilot research studies are critical,” he says, “so that screening is ready to go when treatments are developed.”
Howell, who has specialized in genetic diseases of children since the 1960s, finds this an exciting period.
“We have an approved drug for Pompe’s disease. Newborn screening is in development for SMA, with the anticipation of a treatment. And for Duchenne, there are treatments on the horizon. It’s a remarkable time for someone like me, who’s been in this business a long time.”
Johnson too, believes times are changing.
“There are things that an MDA clinic physician might do differently now in a child with an early, presymptomatic diagnosis of DMD, things that weren’t available during the pilot program, such as maybe starting earlier steroid treatment,” she notes.
But she still has mixed feelings about newborn screening for diseases in which a newborn diagnosis isn’t a clear necessity. Johnson echoed the parents’ belief that, while the newborn period may not be the best time for a diagnosis of muscular dystrophy, that doesn’t mean families wouldn’t benefit from an earlier DMD diagnosis, such as a screening program at age 1 year might provide.
When clinical trials in newborns for DMD become a reality, Johnson says, “it will be very important to revisit MD newborn screening and to carefully explore the best way to provide parents with both information and support.”
Negative DNA Tests
In 1999, when Christian was born to Monette and Albert Smith of Round Rock, Texas, he was gray and floppy, with poor breathing and swallowing efforts. He was sent immediately to the newborn intensive care nursery, where a neurologist diagnosed type 1 SMA, which carries a very poor prognosis.
The Smiths took Christian home, not expecting him to survive for more than about two years. But as the months passed and the Smiths learned more about SMA, “it became more and more evident to me that Christian was not displaying the typical symptoms of the other children and had some other symptoms that none of them had,” Monette Smith recalls.
“It was very difficult for my husband and me to make a decision about whether or not they should trach him [put in a tracheostomy tube] when we didn’t even know what he had.”
After agonizing months spent expecting to lose Christian soon, dealing with medical problems and becoming more confused, the Smiths ended up in the office of neuromuscular disease specialist Susan Iannaccone at the MDA clinic in Dallas, then at Scottish Rite Hospital for Children.
“Dr. Iannaccone was fairly certain after only a 15- to 20-minute clinical examination that Christian did not have SMA,” Smith remembers.
Further testing confirmed that he had nemaline myopathy, which, while a severe disease, carries a better prognosis than SMA1. Christian is now 7 years old.
Ironically, Christian’s doctor had ordered a DNA test for SMA when Christian was born, when the test was fairly new. The test was negative for SMA, but the physician told the Smiths that the test wasn’t 100 percent accurate and that he was certain Christian had SMA1 anyway.
“I agree with genetic screening for newborns in general,” Smith says. “But I feel things could have been handled better and spared my son and my family a lot of unnecessary heartache.
“I felt like the coordination between the doctor who was actually authorized to make the diagnosis for Christian and the genetics specialists who know the most about these disorders was lacking.”
Screening Resources
Editor’s Note: This is a resource list only and implies no endorsement by MDA of these companies or organizations.
Genetic testing (all types)
Genetic tests are now available for many of the diseases in MDA’s program. Prices range from a low of about $260 to a high of $3,800, depending on the specific test and the lab. Insurance coverage is highly variable. Most tests require only a blood sample, from which cellular DNA is extracted.
Most laboratories that do this kind of testing can also test fetal cells, but only a few can do preimplantation testing. See your MDA clinic physician or genetic counselor and/or contact one of the companies listed below.
Athena Diagnostics
Worcester, Mass.
(800) 394-4493, (508) 756-2886
https://www.athenadiagnostics.com/
GeneDx
Gaithersburg, Md.
(301) 519-2100
https://www.genedx.com/
GeneTests
National Institutes of Health and University of Washington Seattle
https://genetests.org
Go to “Laboratory Directory” and enter name of disease.
Preimplantation genetic diagnosis
Genesis Genetics Institute
Applied Genetics Technology Center at Samaritan
Detroit
(313) 544-4006
https://www.coopergenomics.com/
Genetics & IVF Institute
Fairfax, Va.
(800) 552-4363, (703) 698-7355
https://www.givf.com/
Reproductive Genetics Institute
Chicago
(773) 472-4900
https://rgiscience.com/
Editor's note: The original title of this article was Do You Really Want to Know? The Pain and Promise of Prenatal and Newborn Genetic Diagnosis. The title was later changed to The Pain and Promise of Prenatal and Newborn Genetic Diagnosis to better reflect the content of the article.
MDA Resource Center: We’re Here For You
Our trained specialists are here to provide one-on-one support for every part of your journey. Send a message below or call us at 1-833-ASK-MDA1 (1-833-275-6321). If you live outside the U.S., we may be able to connect you to muscular dystrophy groups in your area, but MDA programs are only available in the U.S.
Request Information